| Topics | Approaches |
Animal cells use mechanical forces to sense and configure the environment and to signal each other. Our research is focused on understanding the mechanical sensing and actuation processes, using a combination of biological, physical, chemical, and computational tools. Our projects are positioned at the interface of basic science, engineering, and clinical translation to maximize the impact on areas such as tissue engineering, wound healing, and cancer treatment.
How Cell Migration Responds to Physical Stimuli
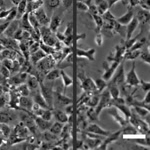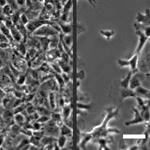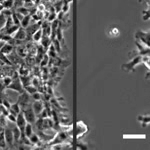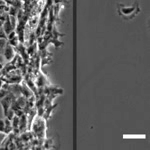 |
We have been investigating the responses of cell migration to physical signals such as stiffness, forces, geometry, and 3D topography. Since the formation of most tissues involves the migration of different cell types to specific locations, the knowledge is important to tissue engineering and repair. In addition to properties like speed, persistence, and directionality, we are examining the initiation of cell migration known as symmetry breaking, and the migration of cell collectives, where the cross-talks between cell-cell interactions and cell migration can lead to the formation of complex structures and macroscopic shapes. |
How Cells Respond to Cyclic Forces
 |
Cells in many parts of the body, including blood vessels and lungs, experience cyclic mechanical forces, which are known to cause profound responses. For example, many cell types show reorientation perpendicular to the direction of cyclic forces, but the mechanism is unclear. We have designed a novel approach for recording the distinct responses during the stretching and relaxation phases of a stretching cycle, under the hypothesis that reorientation is driven by differential retraction or extension along perpendicular directions, possibly in response to stretching and relaxation respectively. In addition, we are testing the hypothesis that the combined responses of cell orientation, cell migration, and cell-cell adhesion may cause cell colonies to undergo rearrangement and morphogenesis-like events. |
Traction Force Microscopy 2.0
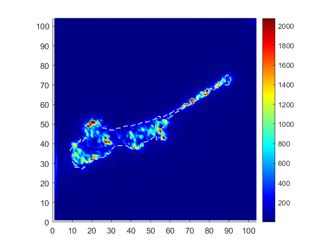 |
Traction force microscopy, as a fundamental tool in mechanobiology, maps the mechanical stress a cell applies to a surface. Cells are plated on elastic substrates embedded with fluorescent microbeads as markers. Linear elastic theory is then used for calculating the distribution of stress based on strains detected with the beads. However, since its introduction more than 20 years ago |
3D Printing of Polyacrylamide: applications from Basic Mechanobiology to Artificial Organs



